 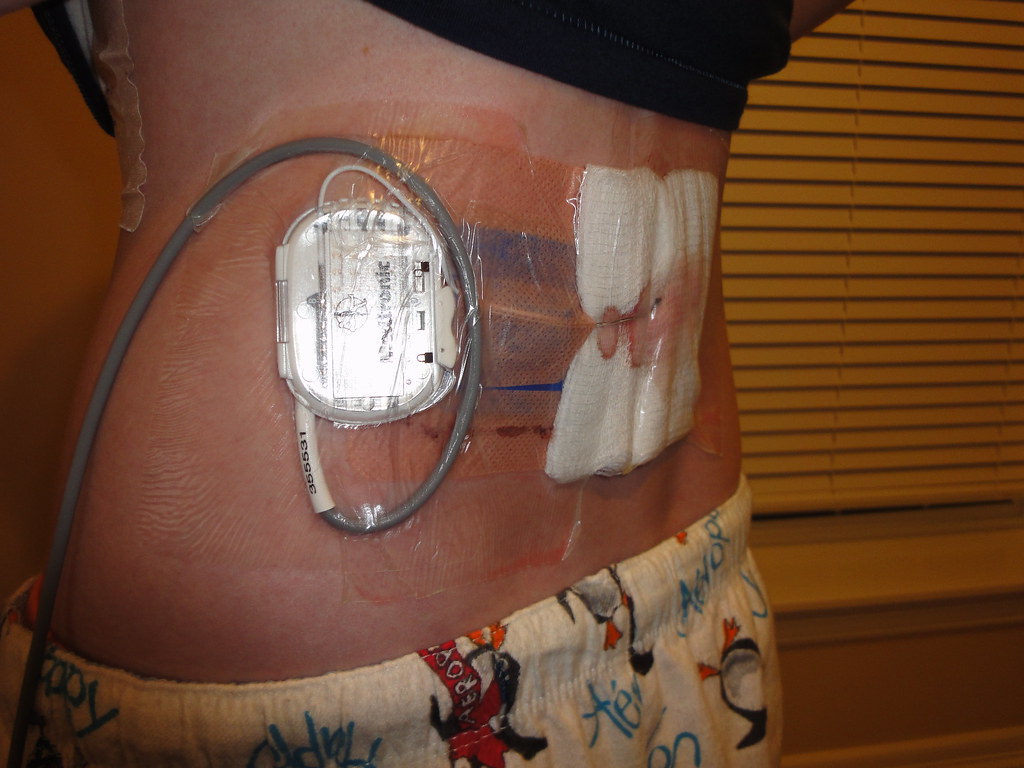
This system is contraindicated for patients who are unable to operate the system or who have failed to receive effective pain relief during trial stimulation. Indications For UseĪbbott Medical spinal cord stimulation (SCS) systems are indicated as an aid in the management of chronic, intractable pain of the trunk and/or limbs, including unilateral or bilateral pain associated with the following: failed back surgery syndrome, nonsurgical back pain (without prior surgery and not a candidate for back surgery), and diabetic peripheral neuropathy of the lower extremities. The system is intended to be used with leads and associated extensions that are compatible with the system. This neurostimulation system is designed to deliver low-intensity electrical impulses to nerve structures. Spinal Column Stimulation (SCS) Systems Intended Use Eterna™ SCS IPG Size Comparison Memo (MAT-2210151). 
Success using neuromodulation with BURST (SUNBURST) study: results from a prospective, randomized controlled trial using a novel burst waveform. Deer T, Slavin KV, Amirdelfan K, et al.Clinical Summaries for Spinal Cord Stimulation Systems Clinician’s Manual. Surgical treatment of refractory low back pain using implanted BurstDR spinal cord stimulation (SCS) in a cohort of patients without options for corrective surgery: findings and results from the DISTINCT study, a prospective randomized multi-center controlled trial. Yue JJ, Gilligan CJ, Falowski S, et al.medical management for low back pain (DISTINCT). Epidemiology of chronic low back pain in US adults: data from the 2009-2010 National Health and Nutrition Examination Survey. ‡ Indicates a third party trademark, which is property of its respective owner. Based off comparison to volumetric measurement of the following IPGs: Boston Scientific ‡ WaveWriter Alpha ‡ 16: 20.1 cc Medtronic ‡ Intellis ‡: 13.9 cc Nevro ‡ Omnia ‡: 26 cc Saluda ‡ Evoke ‡: 33cc. ¶ Smallest size determined by volume in cubic centimeters. Refer to the Instructions for Use for full details on the MR Conditional scan parameters. § BurstDR™ stimulation is superior when compared to traditional tonic stimulation as studied in SUNBURST. † Post-hoc analysis from the data collected in the study. Patient reported outcomes are usually assessed after each patient interaction and therapy adjustment including medication changes made as needed in order to optimize pain control. Interventional therapy such as injections and radio frequency ablation is also allowed. Supervised interventional therapy may include - but is not limited to - physical therapy, chiropractic care, back school, cognitive behavioral therapy and acupuncture. Medication optimization includes use of non-steroidal, anti-inflammatories and muscle relaxants, as appropriate. * Known as CMM during a patient’s follow-up period, subjects will receive supervised medical care, including medication optimization and supervised non-interventional therapy. BurstDR™ Stimulation, patented technology exclusively from Abbott, is also referenced to as Burst stimulation in clinical literature. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
